Use of Vial Adapters Can Reduce Drug Overfill
Vial adapters are easy to use and pose less risk of needlestick injury than traditional syringe and needle manipulation. They not only provide needle-free transfer of liquid for reconstitution, but also enable consistent withdrawal of drug product from a vial. By providing such consistent withdrawal, the use of vial adapters can aid in reducing the amount of required overfill (extra drug product in a vial to address the fact that not all drug product can be removed), which may result in cost savings.
![]()

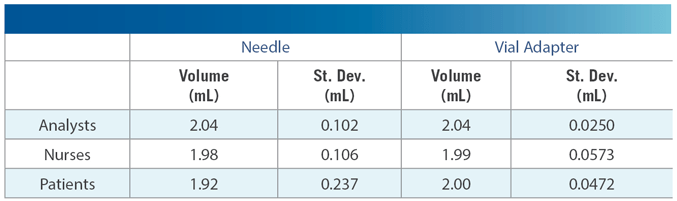
West has conducted a study examining this point: comparing the volumes of liquid (water for injection, or “WFI”) withdrawn from a vial via: (a) traditional needle penetration, and (b) use of a Vial Adapter 13mm female Luer lock (FLL). The study comprised three groups: 4 laboratory analysts experienced with vial adapters, and 20 persons not experienced with vial adapters (10 nurses and 10 persons suffering from hemophilia). Each person was trained on both withdrawal methods. Over 100 measurements were made with each. Data were weights of syringes before and after withdrawal.
*Internal data on file
Use of the Vial Adapter gave better consistency. The standard deviation for a 2 mL withdrawal was substantially lower with the Vial Adapter for all groups (see the table). From this it can be judged that delivery volume to the patient will likewise be more consistent – and with better volume of delivery, it follows that the amount of overfill can be reduced. As noted – this is a benefit to both the patient and to the pharmaceutical manufacturer. For more information visit the website or contact a Technical Customer Support representative.
Vial Adapters are 510(k) cleared by the United States Food and Drug Administration and carry the CE mark (0344). Products are shown for INFORMATION purposes only and may not be approved for marketing in specific regions. Distribution and use are subject to applicable regulatory approvals and requirements for medical devices. Please contact your West Pharmaceutical Services, Inc. (West) representative for product availability.Important product information, safety and warnings are included at: https://www.westpharma.com/products/vial-adapter-systems/vial-adapters




