Important Concerns Regarding Volume and Viscosity Part 2
In Part 1 of this three-part blog series, we reviewed some of the current market trends and challenges specific to biologic drug products, as well as the opportunities within drug delivery for drug products of higher viscosity and greater injection volume.
![]()
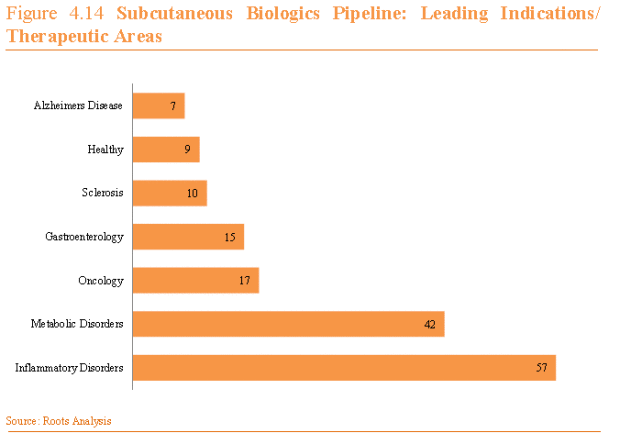
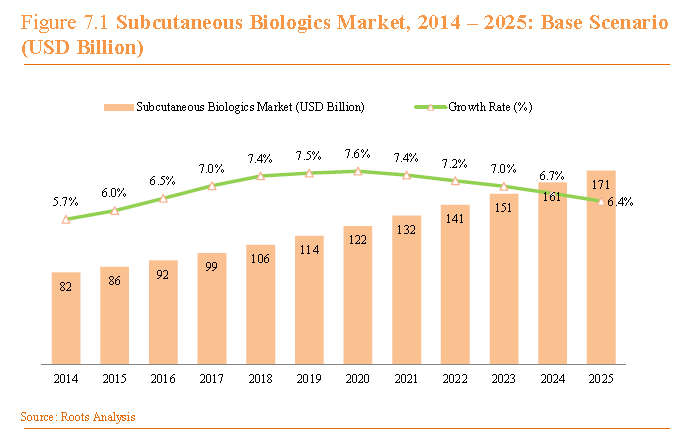
Part 2 will discuss the pipeline and therapeutic areas where larger volume and viscosity are being explored, as well as the impact of these trends to the respective patient population and the overall health delivery system. During the last several years, we have seen an increasing trend toward the self-administration of injectable drugs, with much of the growth coming from the launch of several monoclonal antibodies to treat inflammation (Arthritis, Psoriasis, Crohn’s Disease). Many of these drug products pair a prefilled syringe with an auto-injector to support safe self-use by patients. If we look at the pipeline for products in development for large molecule therapy, many of the therapies under development follow this same trend as the majority of research and development programs targeting drugs to treat inflammation in the form of antibodies. Of 196 subcutaneous biologics in development, 94 are antibodies, representing 48% of the pipeline (Roots Analysis). Other therapeutics areas in the pipeline include metabolic disorders (diabetes, growth hormone deficiency) and oncology (Figure 1). When considering the larger volume injectable market, many of these pipeline drug products will be evaluated in greater than 2mL dosing, but there is also a large opportunity for intravenous (IV) or intramuscular (IM) drug products to move to a subcutaneous delivery system, which expands the potential for larger volume delivery and self-injection. The healthy pipeline, in addition to this new opportunity for IV therapy, will contribute to steady biologics growth over the next several years, as outlined in Figure 2 below from Roots Analysis.
As the market for biologics grows over the next several years, pharmaceutical companies will be tasked with selecting the appropriate delivery device and container for the drug. These decisions can be complex, with injection frequency, dose volume and viscosity, delivery rate and duration, and human factors all contributing to the selection of an appropriate delivery system. In Part 3 of this blog series, we will discuss the various technologies in drug delivery developed specifically for delivery of drug products with larger volume and higher viscosity. We will also assess newer innovations in this market and discuss the advantages and disadvantages relative to both the container and delivery system.




