Overcoming Challenges Associated with Biologic Drug Formulation and Development
Standing front and foremost in biologic drug formulation is the optimization of safety and efficacy while ensuring quality and stability during manufacturing. Biologic manufacturing subjects the formulation to different stresses and conditions that may compromise quality and stability. Recently, the requirement for the development of subcutaneous formulations for high dose drugs, such as monoclonal antibodies, at high protein concentrations has created additional challenges, including degradation.
![]()
Recent formulations have been designed to minimize protein aggregation and decrease viscosity, which has led to successful manufacture of many biologics. Current challenges focus on dealing with the high drug volumes and increased concentration, which get pushed upstream to the drug formulators. The panacea of successful drug formulation would be: liquid stable, <1mL, viscosity of <10cP and an injection time of <15 seconds. These product attributes also need to consider the patient as lack of patient compliance will override any novel development the drug formulators make.
The patient experience can be improved using formulation and delivery device technologies. Impact to the patient experience can be improved by:
- Formulating drugs to higher concentrations which reduces dosing frequency
- Using higher volume delivery systems to deliver a larger volume and reduce dosing frequency
- Utilizing delivery systems which allow drug administration in the home setting thereby avoiding frequent, inconvenient, and often expensive trips to the clinic
Formulating drugs that can be self-administered by the patient have progressed the treatment and compliance of disease states such as rheumatoid arthritis and diabetes, with the fundamental shift being driven by a sub-cutaneous delivery rather than IV or infusion. This move to sub-cutaneous delivery is challenging for formulators: how do you balance the pharmacological needs of the drug with the tolerability of the patient?
There are concerns around the subcutaneous space and its ability to tolerate rapid injection and larger dose volumes. Tissue disruption and site reaction may occur and, if the injection is rapid and the volume too large, there is potential for the product to leak back from the injection site, reducing the bioavailability.
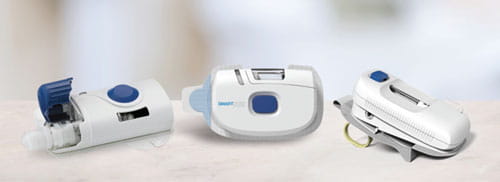
As therapeutic dose volumes increase into the 2-10mL space, wearable drug delivery devices play a crucial part in the solution. A wearable device allows longer dosing times, without negatively impacting patient comfort and convenience, and even allows for home administration. The options provided by wearable technology offers new alternatives that were not previously possible due to historically binding parameters for formulators.
West offers the SmartDose® platform of drug delivery devices to meet a variety of wearable, subcutaneous delivery needs. Click here to learn more about how the platform can enable delivery of your drug product.
SmartDose® is a registered trademark of West Pharma. Services IL, Ltd., a subsidiary of West Pharmaceutical Services, Inc., in the United States and other jurisdictions



