Not All Barriers Are Created Equal
Barriers are very important for drug packaging systems. There are two roles demanded of a barrier. The first is to protect drug products from the environment. This is the primary function of a drug container closure system. The second is to prevent drug products from interacting with the packaging materials themselves. Elastomer closure manufacturers have been working to improve these barriers through multiple technologies. The technologies used on commercial products are traditional coating, and film lamination. Each has pros and cons.
![]()
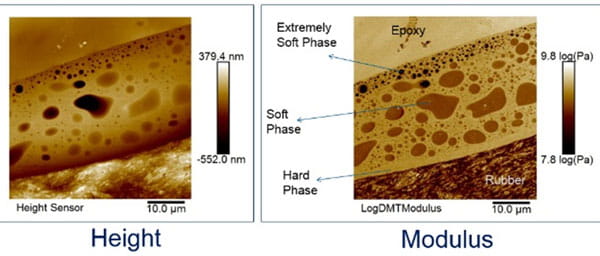
Traditional coating can cover the entire surface of an elastomer closure. However, coating formulations require solvents and other chemicals, such as process aids, for the coating process (dipping or spray) to be performed. This potentially creates extractables / leachables that can migrate into the drug product. Further, if the coating is not in one phase, the barrier property will be compromised, because chemicals can diffuse through the weakest phase. See above the AFM topography and modulus mapping image of a commercial coating that comprises multiple phases.
Film lamination can be used on select areas of an elastomer closure, and provide a better barrier because it is a uniform layer. Properly selected, the extractables / leachables from such a film will be low. However, due to the rigidity of the film, the sealing area cannot be laminated; therefore, unlike traditional coating, coverage is not over the entire surface. Another factor is adhesion between elastomer closure and film. Poor adhesion causes delamination, which could lead to particulate generation, loss of barrier performance, and sterility problems.
Understanding the importance of barriers, West’s Scientific Insight Lab has developed a multi-faceted approach to quantify the composition and performance: Strategy to Understand Surface Chemistry of Elastomeric Components and Their Interaction with Parenteral Drug Products, TR 2018/188. For more on how West can provide support, contact an Account Manager or Technical Customer Service (TCS) representative.



