Transitioning to ISO 80369‑7: Enhancing Safety and Performance in Vial Adapter™ Transfer Devices
Smarter, Safer, Needle-Free Drug Transfer with West’s Vial Adapter™ Transfer Device
Vial adapter™ transfer devices are needle-free solutions designed to simplify the reconstitution and transfer of drug products. They provide a safe and efficient way to prepare injectable medications while helping reduce the risk of needlestick injuries and particulate contamination [1].
Each device features an integrated plastic spike that connects securely to the vial, enabling smooth fluid transfer without conventional needles. This design not only enhances user safety but also supports consistent drug preparation across different healthcare settings, helping clinicians work quickly and confidently [1]. Vial adapter devices are widely used and valued for their ease of use, compatibility, and reliable performance.

Design Changes to Meet ISO 80369-7 Standards
To maintain compliance with regulatory requirements, West has implemented a design update to its Vial Adapter™ products in alignment with ISO 80369-7 “Small-bore connectors for liquids and gases in healthcare applications”- version 2021. This standard now governs connectors used for intravascular and hypodermic systems, ensuring consistency and enhanced safety across the medical device industry.
Note: Please note that the implementation of the ISO 80369-7 standard applies exclusively to US and EU SKUs and is not implemented across all Vial Adapter transfer devices. This update is not applicable to the Mix2Vial® product line. To confirm whether your specific item is affected, please contact your Technical Service Representative.
Previously, West’s Vial Adapter™ transfer device design adhered to ISO 594-1 “Conical fittings with a 6 % (Luer) taper for syringes, needles and certain other medical equipment” and ISO 594-2 “Lock fittings”. As these standards have now been withdrawn and replaced by ISO 80369-7, the update was necessary to ensure that West’s products conform to the new dimensional and performance specifications.
Importantly, this design change does not alter the overall look, performance, or handling of the Vial Adapter™ transfer device. It ensures manufacturing continuity, supports consistent compatibility, and reflects West’s commitment to deliver products that are both regulatory compliant and safe for healthcare use.
Understanding the Shift from ISO 594 to ISO 80369-7
The medical device industry has moved from ISO 594 1/ 2 to ISO 80369 7 as part of a global effort to standardize small bore connector design. At a high level, this transition supports safer, more consistent use of Luer type connectors across healthcare applications by aligning with updated geometry, performance expectations, and application specific requirements. For Vial Adapter™ transfer devices, adopting ISO 80369 7 ensures continued compatibility while meeting the latest international safety and design criteria.
For a more detailed overview including why ISO 594 was withdrawn, how the ISO 80369 family was developed, and its broader impact on medical connectors explore our companion blog: “Understanding the Shift from ISO 594 to ISO 80369: What Medical Device Manufacturers and Partners Need to Know.”
From Old to New: Design Differences and Testing Strategy
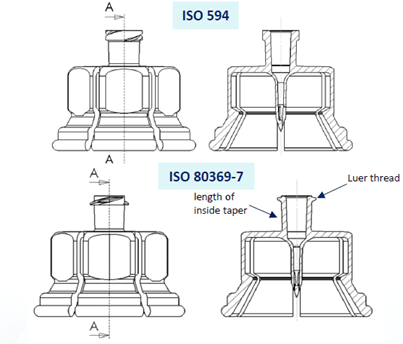
Above picture illustrates the Vial Adapter™ transfer device designs compliant with the former ISO 594 and the new ISO 80369-7 standards. To meet the ISO 80369-7 requirements, the Luer lock thread thickness has been reduced, while the taper angle remains unchanged.
West manufactures Vial Adapter™ products equipped with Luer fittings that fully comply with ISO 80369-7 “Small bore connectors for liquids and gases in healthcare applications, Part 7: Connectors for intravascular or hypodermic applications”. This standard defines the dimensional and performance requirements for connectors used in these applications, ensuring that every product design aligns with global expectations for safety and reliability.
To confirm compliance and maintain product excellence, West conducted a comprehensive series of Design Verification tests for all Vial Adapter™ product families. These evaluations covered both initial (T=0) performance and aged product testing (under accelerated and real time testing in progress), simulating the device’s full shelf life. All verification activities are executed according to West’s established Quality requirements, Product Specifications, and the ISO 80369-7 compliance criteria.
Each Vial Adapter™ transfer device features a Luer fitting on one side and a vial spike and skirt (gripping feature) on the other. Verification testing was conducted holistically to ensure the device performs reliably in all aspects of its intended use. Key testing areas included:
- Luer Connection (as per ISO 80369-7)
- Functional Testing (as per Intended Use Instructions – IFU)
- Vial Connection (no design changes): Although the vial-end design remained unchanged, West performed additional studies to ensure full functional integrity.
- Primary Packaging Validation.
- Secondary Packaging.
All verification results met acceptance criteria defined within West’s verification protocols, confirming that the updated design maintains the same trusted performance, safety, and quality as the previous version, while ensuring full compliance with the latest ISO 80369-7 standard.
Through this design update and comprehensive verification program, West reaffirms its commitment to product quality, patient safety, and regulatory compliance. The transition to the ISO 80369-7 compliant design ensures that the Vial Adapter™ transfer device maintains the same trusted performance and usability customers rely on, while aligning fully with the latest global standards for safe medical device connections.
The Critical Role of Vial Adapter™ Transfer Devices in Drug Preparation and Delivery
As drug manufacturers navigate increasing regulatory expectations, the reliability and consistency of drug delivery components have never been more important. With the transition to an ISO 80369 7 compliant design, Vial Adapter™ transfer devices continue to provide clear, practical benefits for customers across clinical, pharmaceutical, and combination product settings. At the same time, customers gain the advantage of strengthened global compliance.
The ISO aligned design also helps reduce the risk of connection related issues, supporting safe medication handling and helping protect both patients and clinical staff. Because the vial end design remains unchanged, users can rely on the same dependable vial engagement and consistent product integrity throughout the preparation process.
Comprehensive design verification and in house testing ensure that every Vial Adapter™ transfer device configuration maintains the performance, safety, and compatibility customers expect. Early collaboration with West further supports seamless integration into drug delivery systems, helping simplify regulatory pathways and streamline product commercialization.
In a dynamic healthcare landscape, West’s Vial Adapter™ devices continue to enable safe, efficient, needle free drug preparation—enhancing clinical workflows and supporting improved outcomes around the world.
Conclusion
At West, we are proud to partner with our customers to advance the safety and reliability of injectable drug delivery. To learn more about West’s Vial Adapter™ transfer devices or discuss how we can support your next combination product project, reach out to our business development team.
Legal
Vial Adapter™ transfer devices are 510(k) cleared by the United States Food and Drug Administration and carry the CE mark (0344). Products are shown for INFORMATION purposes only and may not be approved for marketing in specific regions. Distribution and use are subject to applicable regulatory approvals and requirements for medical devices. The Vial Adapter transfer device is configurable and may not be suitable for use with all drugs. Please contact your West Pharmaceutical Services, Inc. (West) representative for product availability. Important product and safety information and warnings at: https://www.westpharma.com/-/media/WestPharma/Files/Products/Vial-Adapter-Indications-Safety-and-Warnings.pdf.
This blog is for informational purposes only. West’s products and solutions are sold on the basis that it is the customer’s responsibility to evaluate and test the West product or solution to determine its compatibility with other materials and fitness for any end use. WEST MAKES NO WARRANTIES, WHETHER EXPRESS, IMPLIED OR STATUTORY, INCLUDING, WITHOUT LIMITATION, THE WARRANTIES OF MERCHANTABILITY AND FITNESS FOR PARTICULAR PURPOSE, RELATING TO THE INFORMATION IN THIS DOCUMENT.Vial Adapter is a trademark of West Pharma. Services IL, Ltd., a subsidiary of West Pharmaceutical Services, Inc.
[1] https://www.westpharma.com/products/vial-transfer-devices/vented-swabable-syringe-vial-adapters




