New Technology for Delivery of Vaccines
Vaccines have helped reduce the instance of infectious diseases around the globe, nearly eradicating past plagues, such as smallpox and polio. A very effective method of delivery is intradermal (ID) injection – now made easier with two novel products on the market.
![]()
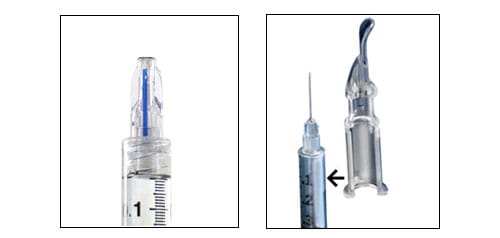
NanoPass MicronJet600™ West's Intradermal Adapter
Intramuscular (IM) and subcutaneous (SC) injections are the most common methods of vaccine delivery. However, the ideal location for injection is the skin. It contains a high concentration of antigen presenting cells that perform an essential role in processing incoming antigens –resulting in powerful immune system responses. Thus, 1ID injection (i.e., delivery to the epidermis or dermis) may result in superior immune responses.
In addition to the superior immune response, ID delivery offers a variety of benefits to pharmaceutical manufacturers, including dose sparing, increased availability of limited or expensive antigens, and reduced cost per dose. Typically, ID injection employs the Mantoux technique, which requires special training, and may not effectively target the skin – resulting either in delivery to the SC tissue (too deep), or leakage (too shallow). These issues of training and inconsistency have deterred many practitioners from using ID injection. To address this problem, West now offers two devices to help deliver a reliable and consistent ID injection each time.
The West ID Adapter device is a snap-on guide for a syringe – ideal for mass vaccinations in developing regions. The MicronJet600™ adapter is a microneedle solution – for a variety of immunotherapies and vaccinations (offered thru our collaboration with NanoPass Technologies, Ltd.) Both devices are suitable for a broad range of ages (2The MicronJet600 was evaluated in various age groups from infants to the elderly)
Contact West today to learn more about how we can help you overcome intradermal delivery challenges.
MicronJet600™ is a trademark of NanoPass Technologies, Ltd.



