Choose Lyophilization Stoppers that Allow Flexibility in Vial Blowback Options
Selecting a container closure system (CCS) is an important part of the pharmaceutical commercialization process. It is important to ensure that the components selected are compatible with the drug product to be packaged; however, it is equally important to ensure that the components are compatible with each other. Here we provide background on the importance of correct pairing between vials and stoppers and showcase lyophilization stoppers V-50-I (13mm) and S-87-I (20mm) in the 4040/40 Gray formulation that can be paired with any vial blowback geometry.
![]()
What parameters affect container closure integrity?
CCS must maintain container closure integrity (CCI) to maintain quality for patient safety. Critical parameters responsible for CCI differ depending on whether the vial is crimped with a seal or not.
Stoppered vials that are not yet crimped with an aluminum seal
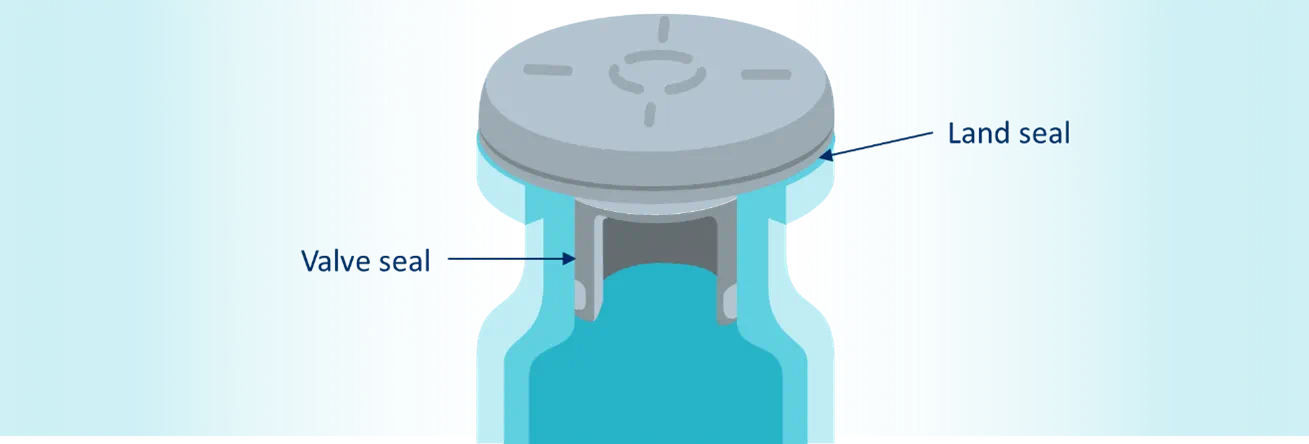
On a fill/finish line, vials will be filled with product and subsequently stoppered either immediately for serum products or within the lyophilization chamber for lyo products. However there is an inherent or potentially unexpected time passing between when the vial is stoppered and when it is capped (whether this is seconds, minutes, or even hours). The stopper must remain fully inserted before capping while ensuring the stopper plug and vial opening interference is capable of maintaining the CCS integrity (see “valve seal ” on Figure 1). When a vial is stoppered, it should be able to achieve an initial level of CCI based on the interference and interaction of the vial neck inner surface and the stopper plug. For this to happen, the stopper must be dimensionally capable to create an interference with the vial opening to achieve CCI and reduce or eliminate the propensity of stopper “pop-up” that could compromise CCI.
Stoppered and crimped vials
The criticality of maintaining CCI only begins during the fill/finish processes. The continuity of maintaining CCI throughout the lifecycle of the drug product is a requirement that is enabled by the crimping of an aluminum seal onto the vial-stopper closure. Once the CCS has been crimped with an aluminum seal, the maintenance of CCI is determined by the seal formed between the underside of the stopper flange and the top of the vial crown (see “land seal” on Figure 1). That seal depends on the heights of the stopper flange, the vial flange, and the seal skirt, as well as the force used to compress and crimp the system.
How do I know if my container closure components will form a compatible system?
The collective component dimensions, component attributes, and fill/finish processes each factor into the success of a CCS. To first assess the capability of a CCS, West’s theoretical dimensional compatibility assessment provides a way to predict whether a given CCS will achieve CCI in two main steps:
- Interference Fit (important for CCI prior to capping)
- Stack-Up Calculation (important for CCI post capping)
An assessment can be performed by a member of the Technical Customer Service (TCS) team, given appropriate product drawings of the vial, stopper, and seal. For West products, the TCS representative will likely already have access to the appropriate information (though may need an item number or preferred design in order to match you with the correct drawing).
Theoretical dimensional compatibility assessment can be divided into two important steps. The first involves assessing whether a given stopper plug and vial opening diameter will be compatible (“Interference Fit”). This assessment will affect the CCI of the system after stoppering, but prior to capping.
To assess the risk to CCI after capping, a stack-up calculation is performed. This involves the analysis of the nominal stopper flange height, vial flange height, and seal skirt length, as well as all tolerances involved. A theoretical compression is assumed, and values are produced for the minimum, maximum, and nominal seal skirt overhang length. If the targeted compression during the fill/finish capping process is known, it is recommended that those values be used within the stack up calculation. If the overhang length is sufficient – 0.5 mm to 1.3 mm – it can be inferred that this container closure system doesn’t have any apparent risks to maintain CCI. However, if the overhang length doesn’t fall within that range, the TCS member will evaluate other possible combinations of components until the necessary overhang is achieved.
Blowback
Neither of these compatibility assessments directly factor in blowback into the equation, however West has shown that CCI can be achieved even if the stopper and vial blowback features are not synchronized, assuming that dimensions are appropriate, and compression is sufficient. This was shown in West studies that are summarized below.
How has West evaluated blowback, and its effects on CCSs?
Effect of Blowback on CCI of Stoppered Uncrimped Vials
West tested lyophilization stoppers V-50-I (13mm) and S-87-I (20mm), formulation 4040/40 paired with ISO standard glass vials with no blowback, EU blowback and US blowback (Technical Report 2019/211 ). These vial – stopper systems were evaluated for raised stoppers and for vacuum retention of uncrimped systems 24 hours after simulated lyophilization process. There were no raised stoppers observed and no significant increase in oxygen level was detected for these samples, indicating successful vacuum retention. West also tested forces required to remove a partially inserted lyo stopper from the vial and force required to fully insert a lyo stopper into a vial for these stoppers and vials with no blowback, EU blowback and US blowback (Technical Report 2019/215). There were no differences between these stoppers across different blowback vials. These results demonstrate that tested lyo stopper designs V-50-I and S-87-I in formulation 4040/40 are universally compatible with US blowback, EU blowback, and no blowback vial configurations.
Effect of Blowback on CCI of Crimped Vials
Lyophilization V-50-I (13mm) and S-87-I (20mm) stoppers, formulation 4040/40 paired with ISO standard glass vials with no blowback, EU blowback and US were then crimped with aluminum seals and CCI was measured via helium leak test method (Technical Report 2019/211). All tested combinations passed CCI.
West also conducted a two-year study to investigate CCI performance of systems incorporating wider combinations of crimped stoppers and vials. Technical Report 2018/199 includes a large variety of stopper formulations, sizes, and configurations; vial sizes, types, and suppliers; compression sets. Stoppers with and without “no-pop rings” were tested with vials with and without blowbacks, and of varying blowback types.
All evaluated systems showed good performance, including the maintenance of CCI over time. Blowback and no-pop rings, or lack of either or both, did not create failure in any of the systems. It can therefore be said that CCI can be achieved regardless of whether a blowback is present or not, for stoppers with and without no-pop rings, as long as dimensions are appropriate, and compression is sufficient.
This compatibility between vials and stoppers is achieved as the inner vial neck – stopper plug surface, where blowback is styled, becomes a secondary seal once a CCS has been crimped. The seal between the stopper and vial flange (see “land seal” on Figure 1) becomes the main responsible surface for CCI, which is why prioritizing the blowback style is no longer seen as a top concern for crimped vials.
How do these findings change how I should approach container component selection?
West recommends a thorough evaluation of dimensional compatibility prior to finalizing container component selections. This can include theoretical compatibility assessments, evaluation of historical data, and/or experimental data of CCI for a system. West can also provide lab services upon selection of components, to evaluate and/or prove CCI for a given system. West studies demonstrated that lyophilization stoppers V-50-I (13mm) and S-87-I (20mm), formulation 4040/40 are capable of maintaining CCI with ISO standard vials before and after vial capping, regardless of the blowback feature.
For more information on this topic, please visit West Knowledge Center (westpharma.com) or contact your Technical Customer Service representative or Account Manager.



