Is Your Legacy Elastomer Item up to the Task?
“Retro.” “Vintage.” “Nostalgic.” These can be great terms to describe that classic car you have your eye on, but they are not great when describing your primary drug product packaging. Many of today’s drug products are still packaged with elastomer formulations and designs that were first developed over 50 years ago. Generic companies that simply replicate the innovator packaging could be affecting their operation negatively for many more years to come.
![]()
Anyone in the generics industry knows that regulatory requirements can change frequently, and almost always to become more restrictive and more complex. The question is: “Can legacy packaging meet all the requirements of today’s ever-changing regulatory landscape?”
Regulation is not the only issue – so is performance. Many years ago, elastomer formulations were designed for operational efficiency. The quality standards were less rigorous and mass production was key. However, as the industry has evolved, formulations improved by employing cleaner raw materials, moving away from latex/dry natural rubber and optimizing designs to improve machinability. Legacy packaging can contribute to machinability failures and higher levels of rejects. Many customers that have learned to “work around” these issues will find it increasingly difficult to do so, as closed systems, such as isolator technology, are more widely adopted. Compounding regulatory and operational issues, legacy packaging components may become unavailable. These supply chain disruptions can put business and patient safety at risk.
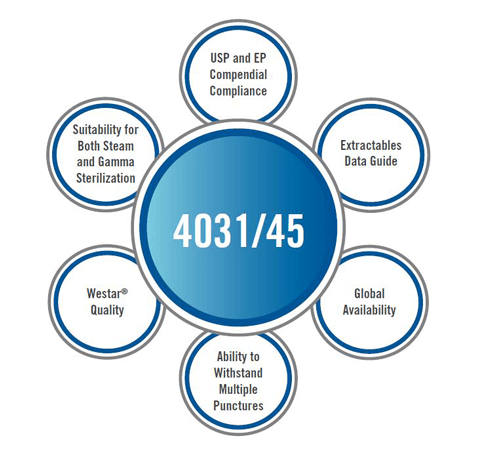
To minimize issues with regulation, performance, and supply chain, it makes sense to switch to a modern elastomer formulation, such as the recently launched West AccelTRA™ 4031/45 gray component. The reauthorization of the Generic Drug User Fee Amendment (GDUFA II) changes planned for October, 2017 eliminate the fee currently associated with filing individual Prior Approval Supplements (PAS)1. This means there is one less barrier faced in order to make primary packaging changes. While making a switch can seem daunting – West can help – making the process much easier through the help of experts in technology and regulatory areas. Living with legacy items is no longer the only option. Contact West today for an evaluation of your current packaging selections as well as identification of any risks present. West can also support with samples, data and/or consultation needed to make the switch.
References:
1. GDUFA II Fee Structure Summary:
http://www.fda.gov/downloads/ForIndustry/UserFees/GenericDrugUserFees/UCM525236.pdf



