Benefits of Electron-Beam Sterilization
Electron-beam irradiation (e-beam - sometimes called beta irradiation) is a technique used commonly to sterilize pharmaceutical packaging products and medical devices. It operates by directing a continuous flow of electrons through the articles being sterilized. Transfer of energy from the e-beam breaks the chains of DNA in organisms, rendering them inactive, and the articles irradiated as sterile. It is used typically for Ready-to-Use (RU) vials and syringes comprising Daikyo Crystal Zenith® cyclic olefin polymer, and Daikyo PLASCAP® seal products.
![]()
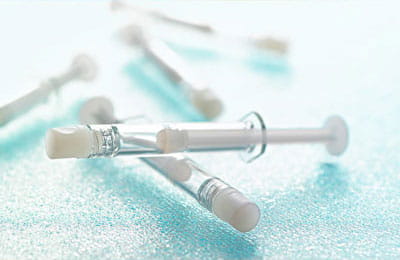
E-beam sterilization has many advantages; it is convenient, gentle and effective. E-beam sterilization can be performed continuously on product in its final package (e.g., product supplied in nests used in flexible fillers). Compared to steam sterilization, it operates at a much lower temperature, and is suitable for products having hard-to-reach areas (e.g., capped cartridges). Compared to gamma irradiation, it poses a much lower risk of degradation (due to shorter exposure times); compared to ethylene oxide exposure, it eliminates the risk of ethylene oxide absorption and subsequent leaching into drug product.
A point to note is that e-beam sterilization can cause a temporary discoloration (yellowish tint) in polymer products, including Daikyo Crystal Zenith® cyclic olefin polymer products. This is due to the presence of short-lived free radicals. However, after the quenching of these radicals, there is no resultant risk to drug product, and in particular, biologic products.
For more information on West products, or how e-beam sterilization can be successfully employed for packaging products, visit the Knowledge Center or contact your Account Manager, or Technical Customer Support representative.
Daikyo Crystal Zenith® and PLASCAP® are registered trademarks of Daikyo Seiko, Ltd.
Daikyo Crystal Zenith® technology is licensed from Daikyo Seiko, Ltd.



